Press Releases
OpRegen® Clinical Data Continues to Demonstrate Functional and Anatomical Improvements in Patients With Dry AMD With Geographic Atrophy
- Eighty-Three Percent of All Cohort 4 Patients Continued to Exhibit Stable or Improved BCVA At Least 6 Months Post-Treatment; Visual Acuity Declined in the Majority of Untreated Eyes
- Three Patients with Evidence of Retinal Tissue Restoration Continue to Demonstrate Areas of Restoration and Sustained Visual Acuity Improvements as of Their Last Clinical Visit
- One Patient with Confirmed Atrophy Growth at Baseline has had Zero Progression for Almost Three Full Years
- Company Intends to Meet with FDA in Q4 2021 to Discuss Clinical Development Plan for OpRegen
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210720005358/en/
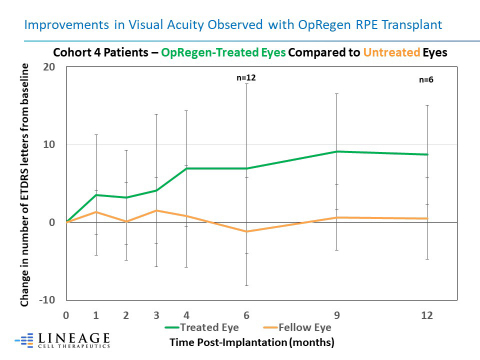
Improvements in Visual Acuity Observed with OpRegen RPE Transplant (Graphic: Business Wire)
“I am excited about the observed changes in visual acuity that show strong trends towards sustained vision improvement in almost all of the treated eyes of Cohort 4 patients as compared to their untreated fellow eyes, which continue to show progression of atrophic AMD and decline of vision,” stated Principal Investigator
“This update further reinforces our view that a suspension of OpRegen RPE cells can generate clinically meaningful anatomical and functional outcomes in patients with dry AMD with GA, particularly in those with earlier-stage atrophic disease,” stated
Overall, 10/12 (83%) of the Cohort 4 patients’ treated eyes continued to be at or above baseline visual acuity at their last assessment, based on per protocol scheduled visits ranging from 6 months to approximately 3 years post-transplant. Improvements in best corrected visual acuity (BCVA) for Cohort 4 patients reached up to +19 letters on the Early Treatment Diabetic Retinopathy Study (ETDRS) chart. In contrast, 10/12 (83%) of the patients’ untreated eyes were below pre-treatment baseline values at the same time points. Among the six Cohort 4 patients treated between September and
Three patients with evidence of retinal restoration and confirmed history of GA growth continue to demonstrate areas of retinal restoration as of their last assessment, ranging from 6 months to approximately 3 years after treatment. Notably, on Optical Coherence Tomography (OCT) analyses, the first Cohort 4 patient with evidence of retinal restoration and confirmed history of GA growth, has demonstrated zero growth in the area of atrophy (GA) almost 3 years following treatment with OpRegen. This is unprecedented due to the progressive nature of the disease. Applying the expected rate of progression based on changes from historical images to the baseline assessment, the size of the atrophic lesion at the visit would have been 4.58 mm using square root transformation (SQRT). Instead, the lesion measured at 2.8 mm SQRT, which was the same as it was at baseline, meaning there was no GA growth over almost 3 years, representing a 63.6% (1.78 mm SQRT) smaller area of GA than was expected. The area of GA in this patient’s untreated fellow eye, which was less severely impacted at baseline, progressed as expected from 3.63 mm to 4.01 mm using SQRT, an increase of 10.5% or 0.38 mm. Notably, microperimetry data collected at the Year 2 and Year 3 post-treatment study visits indicated improvements in the patient’s ability to discern different intensities of light and the patient has experienced clinically significant improvement in visual acuity for more than 2 years, at one point gaining 12 letters on an ETDRS scale. In addition to positive anatomical changes, all three patients with evidence of retinal tissue restoration had visual acuity increases above baseline levels within 9 months post-treatment and visual acuity has remained relatively stable over time compared to their fellow eyes.
Outer retinal layer restoration, which was observed using clinical high-resolution OCT, was evidenced by the presence of new areas of RPE monolayer with overlying ellipsoid zone, external limiting membrane, and outer nuclear layer, which were not present at the time of baseline assessment. These findings suggest integration of the new RPE cells with functional photoreceptors in areas that previously showed no presence of these cells. These effects were most prominent in the transitional areas around the primary area of GA.
These findings of retinal restoration have been confirmed utilizing multiple imaging technologies. The use of multiple imaging modalities differs from traditional assessment of GA progression, which employs only fundus autofluorescence (FAF) to assess changes in the total surface area of the apparent GA over time. Using only FAF may fail to identify structural changes that can be observed only with the addition of OCT imaging. The use of OCT allows for a more precise determination of changes in retinal thickness, organization, and overall health of the retina in areas of potential atrophy, benefits which are possible with cell transplant therapy.
The loss of RPE cells over time creates progressively larger areas of atrophy in the adult retina, leading to impaired vision or complete blindness, a condition known as atrophic AMD. Humans lack the innate ability to regenerate retinal tissue and replace lost retina cells, which led to a presumption that progression of GA may someday be slowed or halted but could not be reversed. The unique findings from the ongoing OpRegen clinical study support a different view, in which an RPE cell transplant can potentially replace or rescue retinal cells in patients who suffer from retinal lesions or degeneration. The totality of these findings supports the view that atrophic AMD is not an irreversible, degenerative condition and that some portion of diseased retinal tissue may be recoverable.
Improvements in Visual Acuity Observed with OpRegen RPE Transplant. The graph represents the mean changes in Best Corrected Visual Acuity (BCVA) for Cohort 4 patients’ treated eye when compared to their fellow eye over a period of 12 months.
About OpRegen
OpRegen is currently being evaluated in a Phase 1/2a open-label, dose escalation safety and efficacy study of a single injection of human retinal pigment epithelium cells derived from an established pluripotent cell line and transplanted subretinally in patients with advanced dry AMD with GA. The study enrolled 24 patients into 4 cohorts. The first 3 cohorts enrolled only legally blind patients with BCVA of 20/200 or worse. The fourth cohort enrolled 12 better vision patients (BCVA from 20/65 to 20/250 with smaller mean areas of GA). Cohort 4 also included patients treated with a new “thaw-and-inject” formulation of OpRegen, which can be shipped directly to sites and used immediately upon thawing, removing the complications and logistics of having to use a dose preparation facility. The primary objective of the study is to evaluate the safety and tolerability of OpRegen as assessed by the incidence and frequency of treatment emergent adverse events. Secondary objectives are to evaluate the preliminary efficacy of OpRegen treatment by assessing the changes in ophthalmological parameters measured by various methods of primary clinical relevance. OpRegen has been well tolerated to date and there have been no new, unexpected ocular or systemic adverse events or serious adverse events that have not been previously reported. OpRegen is a registered trademark of
About Age-Related Macular Degeneration
Age-related macular degeneration (AMD) is an eye disease that can blur the sharp, central vision in patients and is the leading cause of vision loss in people over the age of 60. There are two forms of AMD: dry (atrophic) AMD and wet (neovascular) AMD. Dry (atrophic) AMD is the more common of the two forms, accounting for approximately 85-90% of all cases. In atrophic AMD, parts of the macula get thinner with age and accumulations of extracellular material between Bruch's membrane and the RPE, known as drusen, increase in number and volume, leading to a progressive loss of central vision, typically in both eyes. Global sales of the two leading wet AMD therapies were in excess of
About
Forward-Looking Statements
Lineage cautions you that all statements, other than statements of historical facts, contained in this press release, are forward-looking statements. Forward-looking statements, in some cases, can be identified by terms such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “design,” “intend,” “expect,” “could,” “can,” “plan,” “potential,” “predict,” “seek,” “should,” “would,” “contemplate,” “project,” “target,” “tend to,” or the negative version of these words and similar expressions. Such statements include, but are not limited to, statements relating to the potential benefits of treatment with OpRegen in dry AMD patients with GA, the significance of clinical data reported to date from the ongoing Phase 1/2a study of OpRegen, including the findings of retinal tissue restoration, plans to meet with the FDA in 2021 to discuss OpRegen’s clinical development, and Lineage’s potential to become the pre-eminent allogeneic cell therapy company. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause Lineage’s actual results, performance or achievements to be materially different from future results, performance or achievements expressed or implied by the forward-looking statements in this press release, including risks and uncertainties inherent in Lineage’s business and other risks in Lineage’s filings with the
View source version on businesswire.com: https://www.businesswire.com/news/home/20210720005358/en/
(ir@lineagecell.com)
(442) 287-8963
Solebury Trout IR
(Gogawa@soleburytrout.com)
(646) 378-2949
Nic.johnson@russopartnersllc.com
David.schull@russopartnersllc.com
(212) 845-4242
Source:
